Besides the pounding rains that winter and spring can bring, the most dramatic devastation that this time of the year can inflict on crops is freezing. Ornamental and commercial nursery stock can be absolutely devastated in bad years, suffering severe freezing injuries to buds and shoots. The rain that often follows can then set the stage for a notorious and devastating disease, bacterial canker.
This article is devoted to characterizing key features of freezing damage and how it contributes to bacterial canker, with our goal being to define influencing factors of freeze damage that allow us to mount remedial and preventative programs.

Growers must understand how freeze damage impacts plants on a biological level before they can begin to protect against such damage.
Freezing is a physicochemical event in which matter in a liquid state, such as water, is transformed into a solid phase as temperature decreases. Because ice is less dense than water, as evidenced by the fact that ice floats, the transition from liquid to solid reduces the density of water by about 10%. Accompanying this reduction in density is an increase in volume.
As all living cells have free moisture, the sudden freezing and concomitant expansion of water is disruptive to cells. In many cases, the damage takes the form of physical breakage of delicate chemical bonds within cell membranes, as well as damage to sensitive proteins and enzymes that are necessary to life. This chemical change is irreversible. There is no way to reestablish the functionality of a molecule damaged by freezing. In many cases, this is true of plant cells as well. Physical disruption of the cell and its life-sustaining processes precipitates a chain of events that ultimately lead to death.
However, most plants are known to have ways of acclimating to freezing temperatures. Many plant species that are normally killed by temperatures of 23 to 30 degrees Fahrenheit can be conditioned to withstand temperatures as low as -13 degrees. This conditioning generally takes the form of adapting cell membranes to release water into intercellular spaces, the free space between cells. This increases the concentration of salts and other compounds inside the cells. As you may recall from high school chemistry, increasing the concentration of dissolved substances in water will lower the freezing point. Any child who has tried to make homemade popsicles will have had failures where they only succeeded in making icy slush, while their freezer produced ice cubes without issue.
Transferring free water to the intercellular spaces, then, also transfers the freezing water outside the cell while the cells themselves are more freeze tolerant. Upon thawing, the conditioned membranes will reabsorb the moisture back into the cells and reestablish normal physiological functioning.
The cold conditioning of plants grown in California’s Central Valley area generally requires a gradual decrease in ambient air temperatures down to about 27 degrees. Ideally, this transition should take place over a period of no less than 3 to 4 weeks and provide for a consistent decline in both high and low daytime temperatures. Once temperatures reach 27 degrees, the acclimation should last for at least 7 to 10 days before lower temperatures are experienced. Leading into this period, soil nitrogen levels should be low, while that of phosphorus, potassium and calcium are high. Soil moisture should be less than 50% field capacity, and irrigation avoided. Ideally, pruning should be delayed until initial,
freezing temperatures below 27 degrees are passed. Despite the above precautions, however, sudden temperature drops to 23 degrees, for example, can render the following damage to dormant buds and the sensitive shoot terminus in December.
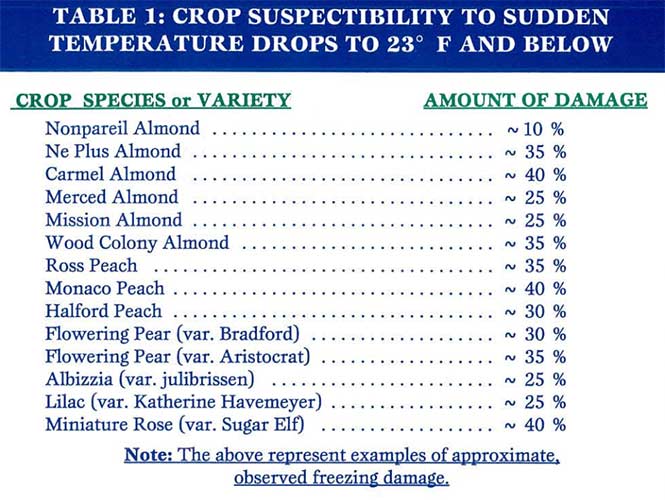 One such freezing scenario is depicted in Table 2. This is not a hypothetical scenario, but one that actually occurred in late 1997, just before the first draft of this article was written and published in our Quantum Yield newsletter.
One such freezing scenario is depicted in Table 2. This is not a hypothetical scenario, but one that actually occurred in late 1997, just before the first draft of this article was written and published in our Quantum Yield newsletter.
 The salient points of this scenario are as follows:
The salient points of this scenario are as follows:
- The month of November was relatively mild and delivered enough rainfall to keep soils moist.
- December 1st through 19th saw relatively mild temperatures, with occasional, interspersed days of lows dropping below 30 degrees.
- December 20th to 30th, daytime temperatures actually increased, while the experienced lows plummeted to below 20 degrees on the 26th and 27th.
The stable daytime highs rendered typical preparations moot, as plants simply were not prepared for the sudden plunge in low temperatures on the 26th and 27th.
Bacterial canker isn’t just a disease that occurs on its own, but rather is often a secondary consequence of freeze damage.
Bacterial canker is a classic predisposition disease in which multiple, contributing factors may be involved. The predisposition characterization is because the primary factor which makes plants vulnerable to canker is freeze damage. All factors that contribute to bacterial canker can be traced back to factors which detract from plant health and the ability of a plant to tolerate or resist freeze damage to tissues. Common elements connected with bacterial canker include:
- Nematode infestations
- Water mold infections
- Mineral deficiencies or excesses
- Irrigation inconsistencies
- Low-lying terrain
- High populations of Pseudomonas syringae on the tissue surface
The integrity of tissues and responsive plant physiology are major characteristics of healthy plants. High integrity of tissues refers generally to the density of such tissues, and more specifically to the stability of the molecular makeup whereby constituent molecules are formed and cross-linked with mineral bridges mediated mostly by calcium, and to a lesser extent, boron. This cross-linking of molecules takes place not only at the cell wall level, but also at the pectin layers (the cement between cells) and on the membranes of both cells and organelles.
For example, elasticity and responsiveness of the membranes to changes in weather patterns is integral to freeze tolerance of plant tissues, much of which is dependent upon the quality of bridging and cross-linkages of phospholipids, glycolipids, cholesterol, and protein molecules with calcium. Insufficient calcium in root tissues, for example, detracts from control of differential permeability, and the tissues begin to leak out cell contents. Eventually, this deficiency in membrane stability and control leads to natural attrition of the affected tissues.
Integrity of membranes is also indirectly necessary to freeze tolerance, as it is the efficient functionality of membranes that leads to efficient photosynthesis and respiration (energy production and transfer reactions of the plant).
Plant nutrition, particularly providing sufficient calcium, potassium and phosphorus, plays a critical role in determining a plant’s resistance to freeze damage and subsequent infections of bacterial canker.
The general role of plant nutrients in bacterial canker resistance can be divided into two major aspects: the effects of sound nutrition on the efficiency of overall metabolism, and the effects of sound nutrition on the structural integrity of tissues. Nitrogen nutrition is of particular importance, as can be seen if we begin this discussion by rating the essential plant nutrients in terms of their availability and ease of absorption from the complex soil environment. A general rating would look like this:
Note that the dichotomy between nitrogen and calcium is the difference between 100 and 5. Recall that one of the most commonly used fertilizers to ‘push’ crops is nitrogen. The more nitrogen that is used, the greater the division between nitrogen and calcium. This is part of the reason why you will commonly hear that high nitrogen fertilization is conducive to disease. That is, the tissues developed from heavy nitrogen applications are those of low integrity and minimal calcium cross-linking of molecules. Plants will not wait for a sufficient harvest of other elements to send forth tissues, but the tissues developed during this interval of nitrogen-aided rapid growth—coincident with severe calcium deficiency—will be much weaker than tissues developed under slower rates of growth.
There are certain varieties of almonds, for example, which host a rapid growth rate (e.g. Padre, Aldrich). Many times, in addition to this existing genetic potential for rapid growth, a grower will further push tree development with nitrogen-based programs, only for trees to start exhibiting severe bark and wood splitting. This is because the rate of tissue growth literally exceeds the rate at which calcium harvest can take place. Many years ago, the forestry industry tried to escalate the rate of growth of various conifers by applying nitrogen fertilization. The trees came up quickly, as expected, but processors noticed that the lumber was splitting to the point of being unusable. The same phenomenon of relative absorption rates was again at play (i.e. N vs. Ca).
One of the more common soil textures conducive to bacterial canker are sandy soils, and typical to sandy soils are low mineral levels of calcium, potassium, phosphorus, and minors, as well as low cation exchange capacity (CEC). Phosphorus is integral to canker resistance, as one of the necessary physiological changes needed for cold acclimation is the elasticity and fluidity of membranes imparted by the presence of unsaturated fatty acids, as opposed to the saturated fatty acid constitution of pho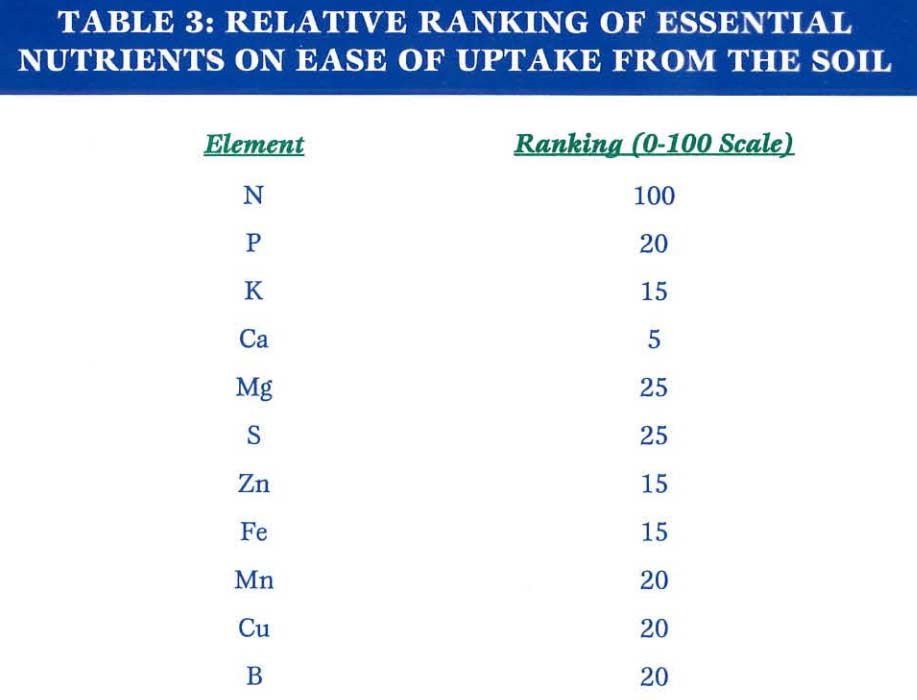 pholipids. Not only is phosphorus needed for the building of phospholipids, but the energy requirement for building phospholipids comes from adenosine triphosphate (ATP), which requires three phosphorus atoms.
pholipids. Not only is phosphorus needed for the building of phospholipids, but the energy requirement for building phospholipids comes from adenosine triphosphate (ATP), which requires three phosphorus atoms.
Potassium, which also lags behind nitrogen harvest, is necessary for metabolism. The efficiency of photosynthesis, for example, relies heavily on absorbed levels of potassium. Potassium is specifically involved as the sole catalyst of over 40 different enzymes and thus the overall metabolism of the plant is very much dependent upon the timely harvest of potassium.
One of the most graphic displays of the lagging of calcium, phosphorus and potassium harvest occurs during the January and February root flush. Roots begin to push, using stored nutrition reserves, but require ongoing mineral harvest as they form. While being able to harvest sufficient levels of nitrogen, again calcium, phosphorus and potassium lag behind. Again, tissue growth does not wait for the sufficient harvesting of these minerals, instead continuing unabated.
Several weeks to a month later, one can excavate the root system and find many newly pushed roots already undergoing natural attrition. Roots will develop tan to dark brown discoloration at the base, the terminus and various parts of the root, all representing points of natural attrition. Natural attrition of the new crop of roots leads to further deleterious events. For one, the tips of the roots are the centers for production of cytokinins, growth hormones which are normally translocated to the growing points to promote cell division and development of tissue stability. Furthermore, the sites undergoing natural attrition become easy targets for infection by water molds, such as Pythium or Phytophthora species. On many occasions, we have observed high incidences of bacterial canker on blocks which host heavy Pythium infections.
Programs that effectively maximize the release of tied-up minerals, and impart natural suppression of both nematodes and various soil-borne pathogens, center around microbial activation of the soil. Over the years, for example, I have observed that the most effective nematode and soil-borne disease control programs emphasize high soil microbial activity, with more than 75% emphasis on immediately addressing growth parameters of the soil, with 25% of attention being directed to gradual, steady debilitation of pest and pathogen populations. To merely address nematode and pathogen control with minimal to no attention to soil growth parameters is both short-lived and generally slower in visible growth response.
Ice nucleation plays a critical role in the development of bacterial canker infections.
Water will oftentimes ‘supercool’ to temperatures well below 32 degrees in the absence of ice formation. This phenomenon is promoted by the absence of a center for ice formation, referred to as ice nucleation.
In 1978, Dr. Steven Lindow of U.C. Berkeley found that certain resident bacteria that normally inhabit the surfaces of plant tissues provided a nucleus for ice formation. In their presence, ice formation took place at 32 degrees. When Dr. Lindow sterilized the plant surfaces and freed them of these bacteria, tissues could be supercooled to 26 degrees before ice formation was induced. It is interesting in that one of the bacterial species found to trigger ice nucleation, Pseudomonas syringae, is also a cause of bacterial canker. Apparently, the polysaccharide coat of these bacteria is endowed with a peculiar molecular arrangement which acts as a starting point for ice formation. In the case of Ps. syringae, the organism facilitates freezing of the host tissue, then seizes the opportunity to colonize the cell contents leaking out of the freeze-damaged cells, before progressing further into the host tissues.
Dr. Lindow surmised that competitively displacing Ps. syringae with another bacterium capable of living on the surface of tissues (called an ‘epiphyte’) could possibly serve as a means of protecting against freezing injuries. He developed a test product, which is now commercially available today in the form of an epiphytic competitive bacterial product called BlightBan.
Many years ago, I also isolated a strain of bacterium from the surface of almond leaves. It was identified as Ps. fluorescens strain 101. The organism was a rapid grower and could effectively displace Ps. syringae from the surface of tissues. We’ve also conducted tests where sterile water was supercooled to 26 degrees without ice formation. The same water holding a droplet of Ps. syringae would freeze at 32 degrees, but water holding the 101 strain of Ps. fluorescens could also be supercooled to 26 degrees.
To ensure more consistent results, I cultured the 101 strain on a unique array of carbon-based food sources for many generations. When time came to spray the 101 onto test trees, the bacterium was sprayed in conjunction with appropriate levels of the carbon food sources to which it was conditioned. The spraying was conducted 2 times at 2-week intervals. About 4 days following the second application, we endured 2 successive days of freezing (approximately 27 degrees during the sensitive stage of nut development). Neighboring blocks suffered greater than 80% freeze damage to the nut crop, while our test blocks only suffered losses of about 8% around the perimeter of the isolated block. Similar levels of protection were achieved with 2 dormant copper applications. This is one of the reasons for applying copper sprays during the dormant season
Nematodes and water molds also warrant examination as contributing factors to bacterial canker.
Various plant-parasitic nematode species can debilitate the root systems of plants. This not only reduces the root surface area for effective absorption of minerals, but oftentimes reduces the sites of cytokinin hormone production. The net consequences of nematode attack are lowered metabolic efficiency, as well as tissue development of low integrity and tolerance to freezing damage. Other researchers, including those at our lab, have conducted “split-root” tests, whereby the roots of host seedlings are split, with some of the split roots then placed into healthy soils, and the other split roots into soil with large populations of either root-knot nematode, lesion nematode, or ring nematode. Another set of plants also have their roots split, but all roots are placed into healthy soil.
Inoculation of the completely healthy soils with doses of Fusarium oxysporum, a vascular pathogen, does not result in disease. However, inoculation of the set with roots placed into nematode-infested soil results in the development of Fusarium wilt. The disease occurs whether inoculation with spores is conducted on the healthy soil or the nematode-infested soil. This means that the predisposition to disease resides not in the physical wounding of tissues exposed to pathogens, but in the overall debilitation of a host’s physiology. That is, the primary effects of nematode infestations are the lowering of metabolic efficiency, loss of photosynthetic harvest, and tissue development of low integrity that is highly susceptible to freeze damage.
The same phenomenon is true of water mold infections of the root system. In fact, we have examined many canker-infected orchards where there were nondetectable to low levels of nematodes, but which hosted damaging populations of Pythium or Phytophthora. Thus, the pre-dispositioning effect of water mold root clipping or debilitation, again, detracts from optimal metabolic functioning and development of sound, freeze-tolerant tissues.
Irrigation practices should also be examined for the role they play in bacterial canker predisposition.
Assuming that the considerations for overwatering and promotion of water mold infections are being addressed, there are some principles of irrigation that deserve coverage. First, stressless irrigation practices are directly linked to the photosynthetic efficiency of plants. This latter factor, the efficiency with which the plant harvests sunlight and converts carbon dioxide to energy and various chemical compounds coincides with our previous concerns about nutrition, nematodes and water molds, and their impacts on this same parameter.
The principle question becomes, “How long should a grower continue to irrigate into the fall season?” Answering this question requires considering the role of irrigation in building tissue integrity and maintaining efficient metabolism. For example, cutting off water prematurely often results in undue stress, which brings about early defoliation, not unlike a heavy infestation of mites. If a late summer or early fall rain should follow, the tree will often respond by attempting to push vegetative buds, thereby expiring buds and energy that are destined for use in the spring push, a weakening event that predisposes the plant to freeze damage and bacterial canker. If a grower should continue to irrigate his plants into late summer, he often runs the risk of tissue succulence and susceptibility to freeze damage, should an early or sudden freeze strike.
Experience has demonstrated that the latter course of action is the preferred path, but it must be combined with nutrition schemes that lean towards low levels of nitrogen and high levels of phosphorus, potassium, calcium and boron. Tissues that develop or are maturing during the latter part of summer and into fall are often fueled by residual fertilizers left over from the season, rather than proactive fertilizer programs. That is, most growers do not institute a conditioning program outside of watering.
Let’s diverge into a related topic. Suppose you are asked to climb Mount Everest, a 29,000-foot-tall mountain, with a professional climber a year from now. One of the first programs you must institute is both physical and physiological conditioning, as one of the greatest obstacles will be the freezing temperatures and low levels of oxygen at high altitudes. To attempt the climb itself is dangerous, but to attempt the climb without rigorous conditioning would be suicidal. Yet, while the Mt. Everest scenario is obvious to all of us, the conditioning of plants for the first frosts oftentimes escapes even the best agriculturists.
Tissues forming in late summer and fall, especially going into winter, must be of equally sound integrity. The shoot terminals are especially prone to development without sound integrity. For example, we see this phenomenon commonly in grapevine canes, with green and immature terminals, which are easily blighted with the first frosts.
The take home message is that we should not relax too soon after harvest. It is the postharvest cultural scheme that sets the stage for the following season. We are often conscientious about tissue mineral status leading up to harvest. However, how many of us take the pains to examine postharvest tissue mineral levels and their ratios? When we talk about frost control, most of us think spring frost control for protection of the current season’s crop. Instead, our first important frost control efforts should be started in the late fall or early winter, because if we can help plants weather the first 2 to 3 frosts, tissues will acclimate and harden off.




